CASE REPORT |
https://doi.org/10.5005/jp-journals-10089-0029 |
Spontaneous Tumor Lysis Syndrome in an Undiagnosed Solid Organ Malignancy: Adenocarcinoma of the Colon
1-3Department of Critical Care Medicine, Manipal Hospitals Whitefield, Bengaluru, Karnataka, India
4Department of Medical Gastroenterology, Manipal Hospitals Whitefield, Bengaluru, Karnataka, India
Corresponding Author: Sanjana Anand, Department of Critical Care Medicine, Manipal Hospitals Whitefield, Bengaluru, Karnataka, India, Phone: +91 7795011297, e-mail: mail.sanjanaanand@gmail.com
Received on: 19 September 2022; Accepted on: 13 October 2022; Published on: 31 December 2022
ABSTRACT
Spontaneous tumor lysis syndrome (TLS) is a rare life-threatening condition resulting from the lysis of a large number of tumor cells. It is more commonly seen in hematological malignancies than in solid tumors. We would like to bring to light the unique case of a 77-year-old gentleman with spontaneous TLS and undiagnosed adenocarcinoma of the colon. He initially presented with spontaneous TLS and was later diagnosed to have metastatic adenocarcinoma of the colon. Given its scarce occurrence and vague clinical presentation, there are very high chances of missing the diagnosis. Early recognition and prompt intervention would significantly reduce morbidity and mortality.
How to cite this article: Anand S, Rajavardhan R, Shetty RM, et al. Spontaneous Tumor Lysis Syndrome in an Undiagnosed Solid Organ Malignancy: Adenocarcinoma of the Colon. J Acute Care 2022;1(2):86-88.
Source of support: Nil
Conflict of interest: None
Keywords: Adenocarcinoma colon, Critical care, Oncological emergency, Solid organ malignancy, Spontaneous tumor lysis syndrome, STLS, TLS, TLS in solid organ malignancy, Tumor lysis syndrome.
INTRODUCTION
Tumor lysis syndrome is an oncological emergency that is characterized by hyperuricemia, hyperkalemia, hyperphosphatemia, and hypocalcemia due to the destruction of numerous rapidly proliferating neoplastic cells. It is usually seen within 1–5 days of chemotherapy induction of hematological malignancies and, rarely, with solid tumors. Occasionally, spontaneous necrosis of malignancies can lead to TLS. Chemotherapy destroys the malignant cells and leads to increased serum uric acid levels from the turnover of nucleic acids leading to kidney failure. Hyperphosphatemia occurs due to the release of intracellular phosphate by tumor lysis, resulting in a reciprocal depression in serum calcium. Hyperkalemia occurs due to the massive destruction of malignant cells. It can also lead to ventricular arrhythmias and sudden death. The mortality rates of spontaneous TLS from solid organ tumors are greater than that from hematological malignancies.1 To date, there have been very few case reports on spontaneous TLS associated with adenocarcinoma of the colon.1-3
CASE DESCRIPTION
A 77-year-old gentleman who is a known case of diabetes mellitus, hypertension, and ischemic heart disease presented with generalized weakness for 1 month, breathlessness, and cough for 1 day. On arrival at the emergency room, the patient was found to have tachypnea, tachycardia, and hypotension. He was started on vasopressor infusion, noninvasive ventilation with oxygen, and shifted to the intensive care unit (ICU). Two-dimensional echocardiography was done and showed signs of pulmonary arterial hypertension. Arterial blood gas was suggestive of severe metabolic acidosis. Serum levels of calcium, phosphorus, uric acid, and potassium were deranged and were satisfying the Cairo–Bishop criteria for TLS.4 Liver function tests were suggestive of ischemic hepatitis as transaminases were markedly elevated and lactate dehydrogenase (LDH) levels were high [alanine aminotransferase (ALT) to LDH ratio of <1.5] for which he was started on N-acetyl cysteine infusion. The coagulation profile was deranged as well. Serum creatinine was found to be slightly elevated to 1.68 mg/dL. The patient was oliguric and also had severe metabolic acidosis suggestive of acute kidney injury (AKI). The patient was not on any regular medication that would cause AKI, hyperuricemia, and hyperphosphatemia. All other causes of AKI and hyperuricemia were excluded. Blood cultures and urine cultures were sent and were found to be negative, thus ruling out sepsis (Table 1).
| Parameter | Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 |
|---|---|---|---|---|---|---|
| Potassium | 5.5 mmol/L | 3.6 mmol/L | 3.3 mmol/L | 3.4 mmol/L | ||
| Uric acid | 11.9 mg/dL | 5.9 mg/dL | 1.8 mg/dL | |||
| Phosphorus | 7.1 mg/dL | 4.4 mg/dL | ||||
| Calcium | 7.5 mg/dL | 8.9 mg/dL | 8.8 mg/dL | |||
| Magnesium | 1.9 mg/dL | 1.7 mg/dL | 2.3 mg/dL | |||
| LDH | 8740 IU/L | |||||
| TLC | 19,800/cu mm | 14,700/cu mm | 9800/cu mm | |||
| AST | 2730 IU/L | 4272 IU/L | 1601 IU/L | 505 IU/L | 270 IU/L | 197 IU/L |
| ALT | 4590 IU/L | 5650 IU/L | 2562 IU/L | 1441 IU/L | 1082 I/L | 810 IU/L |
| APTT | 29.1 seconds | 50.4 seconds | 31.1 seconds | 29.2 seconds | ||
| PT/INR | 17.4 seconds/1.44 | 24.1 seconds/2.06 | 20.7 seconds/1.75 | 18.4 seconds/1.53 | 14.1 seconds/1.15 | 14.2 seconds/1.16 |
| Hemoglobin | 6.2 gm/dL | 8.8 gm/dL | 10.1 gm/dL |
Computed tomography (CT) thorax and abdomen were done and showed multiple hypodense lesions in the liver and scattered multiple nodular lesions in the upper and lower lobes of the lungs suggestive of metastasis (Figs 1 and 2). Asymmetric wall thickening was also noted in the cecum and ascending colon, suspicious of primary malignancy (Figs 3 and 4).

Fig. 1: CT thorax showing scattered mildly enhanced nodular lesions in both upper and lower lobes of the lungs with bilateral moderate pleural effusion

Fig. 2: Contrast CT abdomen and pelvis showing small hypodense lesions in the liver with a mild perihepatic fluid collection
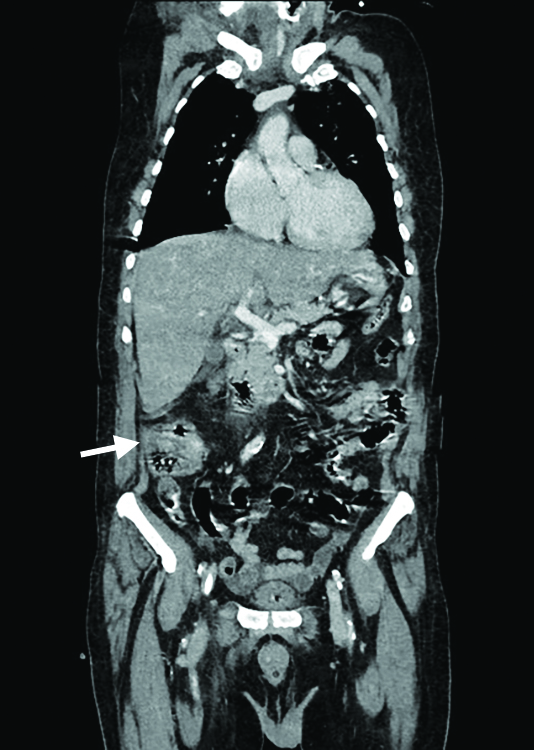
Fig. 3: Asymmetric thickening in ascending colon suspicious for primary malignancy
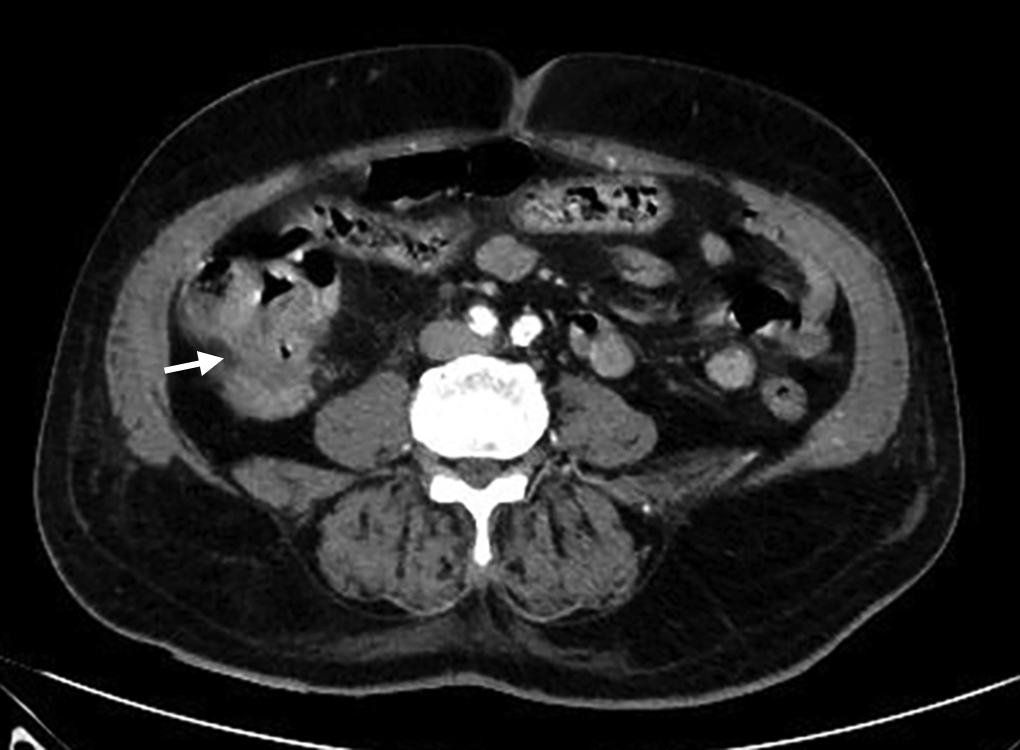
Fig. 4: Asymmetric thickening in ascending colon suspicious for primary malignancy
The patient was immediately started on continuous renal replacement therapy (CRRT) in view of spontaneous TLS and AKI. The patient’s condition gradually improved, and he was eventually tapered off vasopressors and CRRT. Liver function test parameters improved gradually, and coagulopathy got better. Carcinoembryonic antigen level was elevated—8.3 ng/mL. Once the patient got stabilized, a colonoscopy was performed, and the patient was found to have ulceroproliferative growth in the ascending colon with significant luminal narrowing (Fig. 5). Multiple biopsies were taken, and the histopathological examination was conclusive of adenocarcinoma of the colon. Retrospectively on detailed history taking, he did not have typical symptoms of colonic malignancy like altered bowel habits or rectal bleeding, or obstructive symptoms but had significant weight loss and easy fatigability due to severe anemia (probably secondary to gastrointestinal loss). The final diagnosis of metastatic adenocarcinoma of the colon with spontaneous TLS was made. The patient was later transferred out of the ICU once he was stabilized and was then managed on an outpatient basis for the adenocarcinoma.

Fig. 5: Ulceroproliferative growth in ascending colon with critical luminal narrowing
DISCUSSION
This is a case report on spontaneous TLS in an undiagnosed case of adenocarcinoma of the colon. TLS presents with a variety of metabolic abnormalities including hyperuricemia, hyperkalemia, hyperphosphatemia, and hypocalcemia. AKI is one of the most common and serious complications of TLS.5 The first case of spontaneous TLS was reported in the year 1977 by Crittenden and Ackerman in a 50-year-old man with widespread adenocarcinoma of the gastrointestinal tract.6 Spontaneous TLS constitutes almost 15% of all TLS.7 The Cairo–Bishop criteria are used for the diagnosis of TLS4 (Table 2).
| Variable | Value | Change from baseline |
|---|---|---|
| Uric acid | >8 mg/dL | 25% increase |
| Potassium | >6 mg/dL | 25% increase |
| Phosphorus | >4.5 mg/dL | 25% increase |
| Calcium | <7 mg/dL | 25% decrease |
Laboratory TLS
More than two of the following (mentioned in Table 2) should be present.
Clinical TLS
Laboratory TLS with one of the following:
-
Serum creatinine >1.5 times the upper limit,
-
Cardiac arrhythmia or sudden death,
-
New-onset seizures.
Management of TLS includes correction of metabolic derangements as well as aggressive hydration.8 Conventional correction of hyperkalemia, oral phosphorus binders for hyperphosphatemia, as well as for hypocalcemia, allopurinol, and rasburicase for hyperuricemia can be used in the management of TLS.4,9 If conservative measures fail to correct the electrolyte abnormalities or if the patient has low urine output, dialysis should be initiated.9 AKI can prevent timely cancer treatment as well. Therefore, prevention and timely management of AKI are essential in cases of TLS.10
CONCLUSION
Our case report is based on the occurrence of spontaneous TLS which led to the diagnosis of adenocarcinoma of the colon. In this case, TLS was not an anticipated diagnosis as the patient was neither diagnosed with any malignancy earlier nor was the patient on any chemotherapy. Yet our diagnosis was made on the clinical presentation as well as supportive laboratory and radiological evidence which led us toward identifying the primary tumor. Patients with a large tumor burden and with a high cell turnover are at an increased risk of developing spontaneous TLS. Though the incidence is rare in cases of solid tumors, its occurrence can lead to serious complications that can be life-threatening. Hence, early intervention and prompt management are crucial in improving the outcome in these cases.
ORCID
Sanjana Anand https://orcid.org/0000-0003-2465-9988
Rajavardhan R https://orcid.org/0000-0001-9521-2062
Rajesh Mohan Shetty https://orcid.org/0000-0002-9426-6701
REFERENCES
1. Sommerhalder D, Takalkar A, Shackelford R, et al. Spontaneous tumor lysis syndrome in colon cancer: a case report and literature review. Clin Case Rep 2017;5(12):2121–2126. DOI: 10.1002/ccr3.1269
2. Frestad D, Perner A, Pedersen UG. Acute onset and rapid progression of multiple organ failure in a young adult with undiagnosed disseminated colonic adenocarcinoma. BMJ Case Rep 2014;2014:bcr2014205002. DOI: 10.1136/bcr-2014-205002
3. Kalmbach KE, Rahmat LT, Wos JA, et al. A rare oncologic emergency: spontaneous tumor lysis syndrome in metastatic colon adenocarcinoma. Clin Pract Cases Emerg Med 2019;3(4):398–400. DOI: 10.5811/cpcem.2019.9.43770
4. Cairo M, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol 2004;127(1):3–11. DOI: 10.1111/j.1365-2141.2004.05094.x
5. Hsu HH, Huang CC. Acute spontaneous tumor lysis in anaplastic large T-cell lymphoma presenting with hyperuricemic acute renal failure. Int J Hematol 2004;79(1):48–51. DOI: 10.1007/BF02983533
6. Crittenden D, Ackerman GL. Hyperuricemic acute renal failure in disseminated carcinoma. Arch Intern Med 1977;137(1):97–99. DOI: 10.1001/archinte.137.1.97
7. Kekre N, Djordjevic B, Touchie C. Spontaneous tumor lysis syndrome. CMAJ 2012;184(8):913–916. DOI: 10.1503/cmaj.111251
8. Mirrakhimov AE, Ali AM, Khan M, et al. Tumor lysis syndrome in solid tumors: an up to date review of the literature. Rare Tumors 2014;6(2):5389. DOI: 10.4081/rt.2014.5389
9. Coiffier B, Altman A, Pui C, et al. Guidelines for the management of pediatric and adult tumor lysis syndrome: an evidence-based review. J Clin Oncol 2008;26(16):2767–2778. DOI: 10.1200/jco.2007.15.0177
10. Thiery G, Azoulay E, Darmon M. Preventing acute renal failure is crucial during acute tumor lysis syndrome. Indian J Crit Care Med 2007;11(1):29–35. DOI: 10.4103/0972-5229.32434
________________________
© The Author(s). 2022 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and non-commercial reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.